|
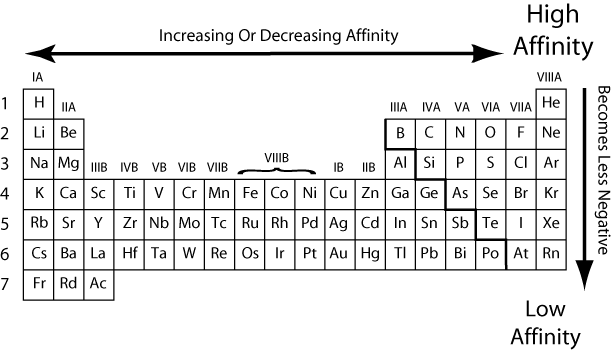
The electronegativity of an element is higher when that element has a stronger attracting ability. In contrast, electron affinity is measured in kJ/mol. Some of the key differences between electronegativity and electron affinity are listed below.Įlectronegativity is the property that causes the electron to be drawn to the atom. Electron affinity, on the other hand, is concerned with the energy released when an electron is added to an atom.Įlectronegativity is a qualitative characteristic, whereas electron affinity is a quantitative characteristic.Īlthough electronegativity is a unitless quantity, it is defined in terms of Pauling.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
